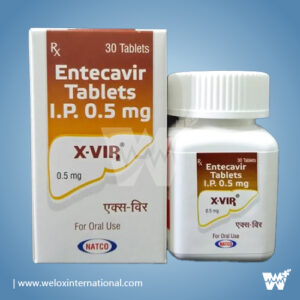
MyHep LVIR™ is a licensed generic of Harvoni® produced in an FDA approved manufacturing facility in India. It’s a direct-acting antiviral used to treat chronic Hepatitis C (HCV) genotypes 1, 4, 5 & 6 in patients with or without liver cirrhosis. For most patients, 12 weeks of treatment is needed to reach cure.
- Harvoni®’s overall established cure rate in patients with HCV genotypes 1, 4, 5, and 6 is roughly 92%
- Harvoni® is not suitable for patients with genotypes 2 or 3. If patients did not get a genotype test, they should be treated with Epclusa® instead
- For treatment-experienced patients or those with advanced Liver cirrhosis (decompensated), the treatment duration is 24 weeks instead of 12 weeks
- Harvoni®’s side effects profile is quite mild, especially with enough water intake
- Consult a doctor before concurrent administration of other drugs or supplements to avoid drug interactions
- The FDA and EMA approve Harvoni® for use in adults and children aged 3 years and older
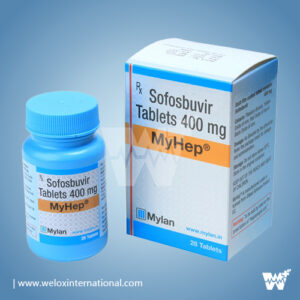
Bottle Size : 28 Tablets (4 weeks supply)
Active Ingredients : Sofosbuvir 400 mg / Ledipasvir 90 mg
Manufactured by Mylan Pharmaceuticals®
Product of India







Reviews
There are no reviews yet.